The Elephant’s Toothpaste Experiment is a highly engaging chemistry experiment that introduces students to catalysts, decomposition reactions, and exothermic energy release. In this supervised science laboratory experiment, students observe how hydrogen peroxide can break down rapidly and create a dramatic foam overflow when a catalyst is added. It is an exciting way to make chemical change visible, memorable, and easier to understand in the classroom.
📘 Overview
Chemical reactions are essential to the transformations that take place in both nature and industry. In a laboratory setting, students can observe how substances break down, form new products, and release or absorb energy. This experiment formed part of a wider study of chemical reactions and equations, helping students connect chemistry concepts to real laboratory observations and everyday life. In the original lab context, the broader lesson also aimed to help students classify reaction types, understand factors that affect reaction rates, and analyze energy changes in chemical reactions.
This specific activity focuses on Elephant’s Toothpaste, also referred to as Lava Falls – Elephants’ Toothpaste. It is a popular demonstration of a rapid decomposition reaction that produces an impressive amount of foam. Through this experiment, students observe the decomposition of hydrogen peroxide into water and oxygen, catalyzed by potassium iodide or yeast, and see how the rapid release of oxygen gas creates a large foam structure.
🎯 Learning Objective
- Students will observe and identify evidence of a chemical reaction.
- Students will understand how a catalyst affects reaction speed.
- Students will recognize the characteristics of a decomposition reaction.
- Students will identify signs of an exothermic reaction.
- Students will connect the experiment to broader ideas in chemical reactions, including reaction rate, gas production, energy change, and laboratory safety.
🧪 Materials

- Hydrogen peroxide (H₂O₂)
- Concentration may vary depending on the level of the activity. Lower concentration creates a slower, safer reaction, while stronger concentrations produce a more dramatic effect and require stricter precautions.
- Concentration may vary depending on the level of the activity. Lower concentration creates a slower, safer reaction, while stronger concentrations produce a more dramatic effect and require stricter precautions.
- Catalyst
- The catalyst may be a potassium iodide (KI) solution or dry yeast mixed with warm water. Either can be used to speed up the decomposition of hydrogen peroxide.
- The catalyst may be a potassium iodide (KI) solution or dry yeast mixed with warm water. Either can be used to speed up the decomposition of hydrogen peroxide.
- Liquid dish soap
- Used to trap the oxygen gas released during the reaction and form the foam.
- Used to trap the oxygen gas released during the reaction and form the foam.
- Food coloring (optional)
- Added for visual effect.
- Added for visual effect.
- Graduated cylinder, plastic bottle, or a volcano model
- Used to contain the reaction mixture.
- Used to contain the reaction mixture.
- Tray or plastic bin
- Used to catch the overflowing foam and contain the mess.
- Used to catch the overflowing foam and contain the mess.
- Safety equipment
- Includes lab coat, safety goggles, and gloves.
📝 Procedure
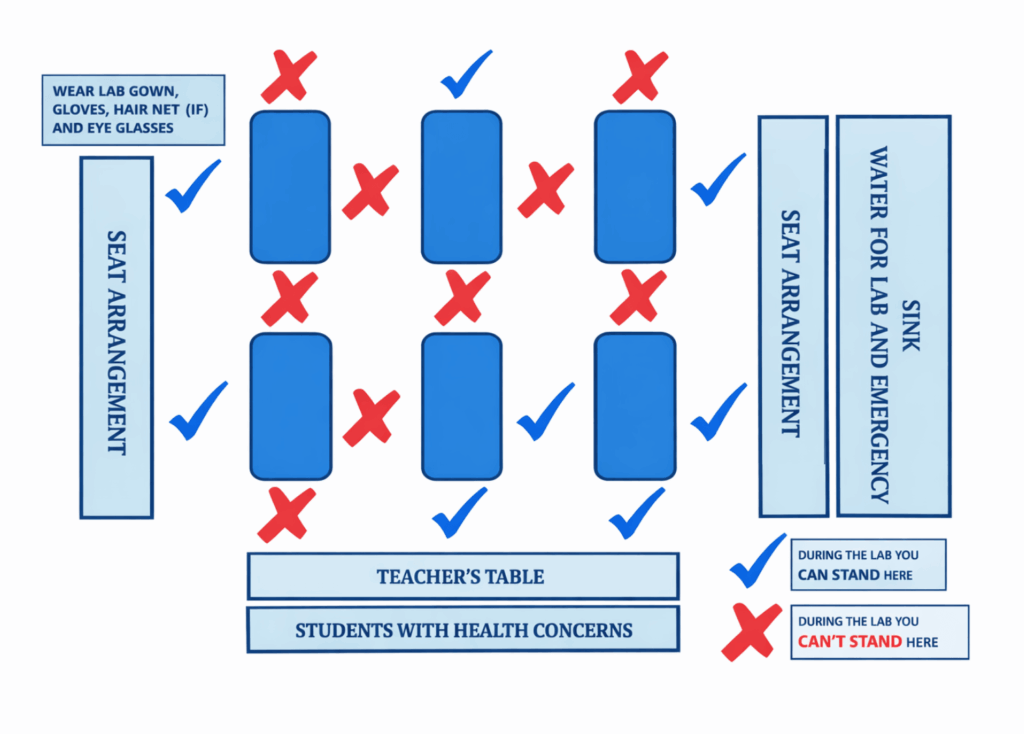
- Ensure all students and the teacher are wearing appropriate safety gear, including safety goggles, gloves, and lab coats.
- Carry out the experiment in a well-ventilated area, especially if stronger hydrogen peroxide is being used.
- Place the graduated cylinder or reaction container in the center of a tray or plastic bin to catch any overflow.
- Add the hydrogen peroxide to the container.
- Add a generous amount of liquid dish soap and gently swirl to combine.
- Add a few drops of food coloring if a more vivid visual effect is desired.
- Prepare the catalyst solution. This may be a potassium iodide solution or a yeast mixture, depending on the setup.
- Carefully add the catalyst to the hydrogen peroxide mixture.
- Step back immediately and allow students to observe the reaction.
- Watch as thick foam rapidly expands and spills out of the container.
- Students should observe the foam formation, heat production, and any color changes as the reaction proceeds.
- Use these observations to discuss gas production, reaction speed, and energy release.
👀 Expected Observations
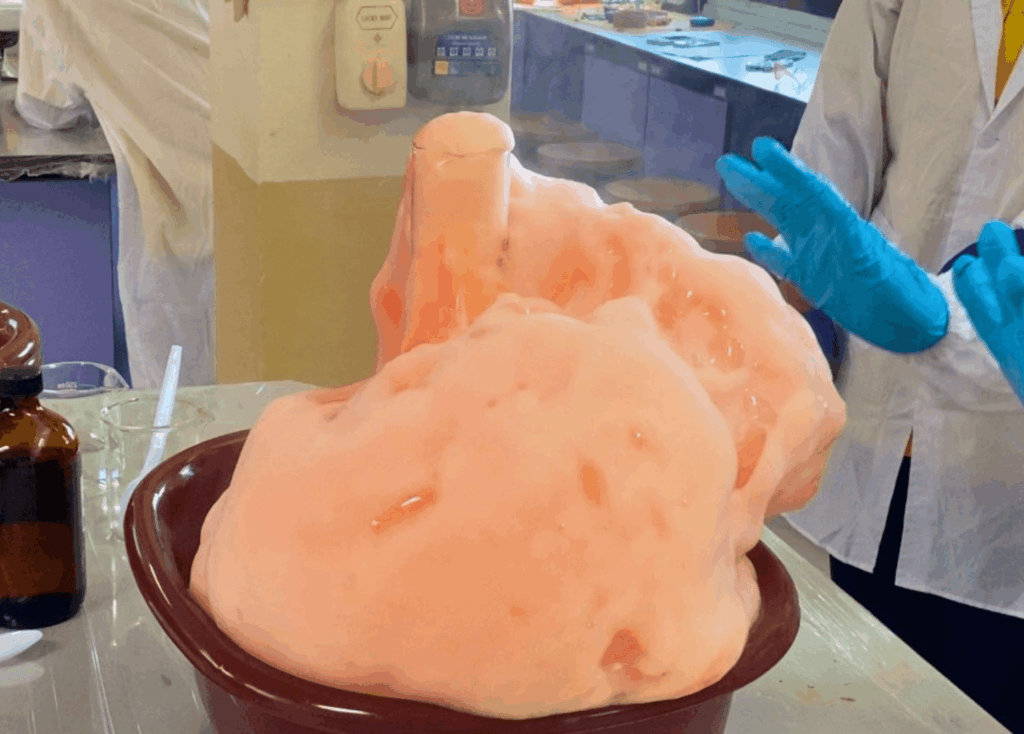
Students may observe:
- Thick foam forming very quickly
- A column of foam overflowing from the container
- A warm container, showing that heat is released
- A visible example of gas production
- Color changes if food coloring is used
- A dramatic and memorable visual effect that resembles toothpaste being squeezed out of a tube
🧠 What’s Happening?
This experiment is an example of a decomposition reaction. Hydrogen peroxide breaks down into water and oxygen gas. On its own, this breakdown can happen slowly over time, but the catalyst speeds it up dramatically. The rapid production of oxygen is what drives the visible reaction.
The dish soap traps the oxygen gas as it forms, creating the thick foam that gives the experiment its name. At the same time, the reaction releases heat, which makes it exothermic. This helps students understand how catalysts can affect reaction speed, how gases occupy space, and how some reactions release energy in noticeable ways.
🌟 Learning Outcomes
Students can learn several important chemistry concepts through this experiment:
- Catalysis
- Students see how a catalyst speeds up a reaction without being consumed in the process.
- Students see how a catalyst speeds up a reaction without being consumed in the process.
- Decomposition Reactions
- Students observe hydrogen peroxide breaking down into simpler substances: water and oxygen gas.
- Students observe hydrogen peroxide breaking down into simpler substances: water and oxygen gas.
- Exothermic Reactions
- Students recognize that the reaction releases heat.
- Students recognize that the reaction releases heat.
- Gas Production and Pressure
- Students understand how rapidly produced oxygen gas creates expansion and foam overflow.
- Students understand how rapidly produced oxygen gas creates expansion and foam overflow.
- Chemical Safety and Experimentation
- Students learn why careful handling, protective equipment, and proper supervision are essential, especially when working with reactive chemicals.
🎓 Classroom Notes
- This experiment works well as a supervised student laboratory experiment when the teacher decides the setup is appropriate for the class and safety conditions.
- In some cases, it may be better presented as a teacher-led demonstration, especially when stronger chemicals or more dramatic setups are involved.
- It fits especially well in a lesson on chemical reactions, decomposition, catalysts, gas production, or energy change.
- The strongest teaching value comes from guided observation and scientific explanation.
- Students can be asked to identify evidence of chemical change, explain why the foam forms, and describe the role of the catalyst.
- This experiment also supports discussion on how chemistry connects to real-world reactions and why lab safety matters.
💬 Discussion Questions
- What signs showed that a chemical reaction took place?
- Why is this experiment considered a decomposition reaction?
- What role does the catalyst play in the experiment?
- Why does the foam rise so quickly?
- Why is this reaction considered exothermic?
- What gas is being produced during the reaction?
- How does the dish soap affect the result?
- What would happen if no catalyst were added?
- How does this experiment help us understand reaction speed?
- Why is safety important when working with hydrogen peroxide?
🚀 Extension / Challenge
- Compare this experiment with another decomposition reaction.
- Research the meaning of catalyst and provide another example from daily life.
- Explain why oxygen gas creates foam when soap is present.
- Create a concept map using the terms catalyst, decomposition, oxygen, foam, and exothermic.
- Write a short explanation of how the experiment demonstrates a chemical change.
- Compare this experiment with the Gummy Bear Explosion Experiment in terms of visible results and energy release.
- Identify three pieces of evidence that show new products formed.
- Explore how the concentration of a reactant can affect reaction rate.
- Describe how this experiment supports learning in a chemical reactions unit.
- Reflect on why visually dramatic experiments can help students remember scientific concepts.
⚠️ Safety Note
This experiment involves reactive chemicals and can generate a rapid overflow of foam and heat. It should only be carried out in a controlled laboratory setting under direct teacher supervision with full protective equipment and proper ventilation. Students should not attempt to recreate this activity on their own or outside a supervised lab environment.
📂 Media & Resources
For downloadable files and complete teaching resources, visit our Download Center.


















